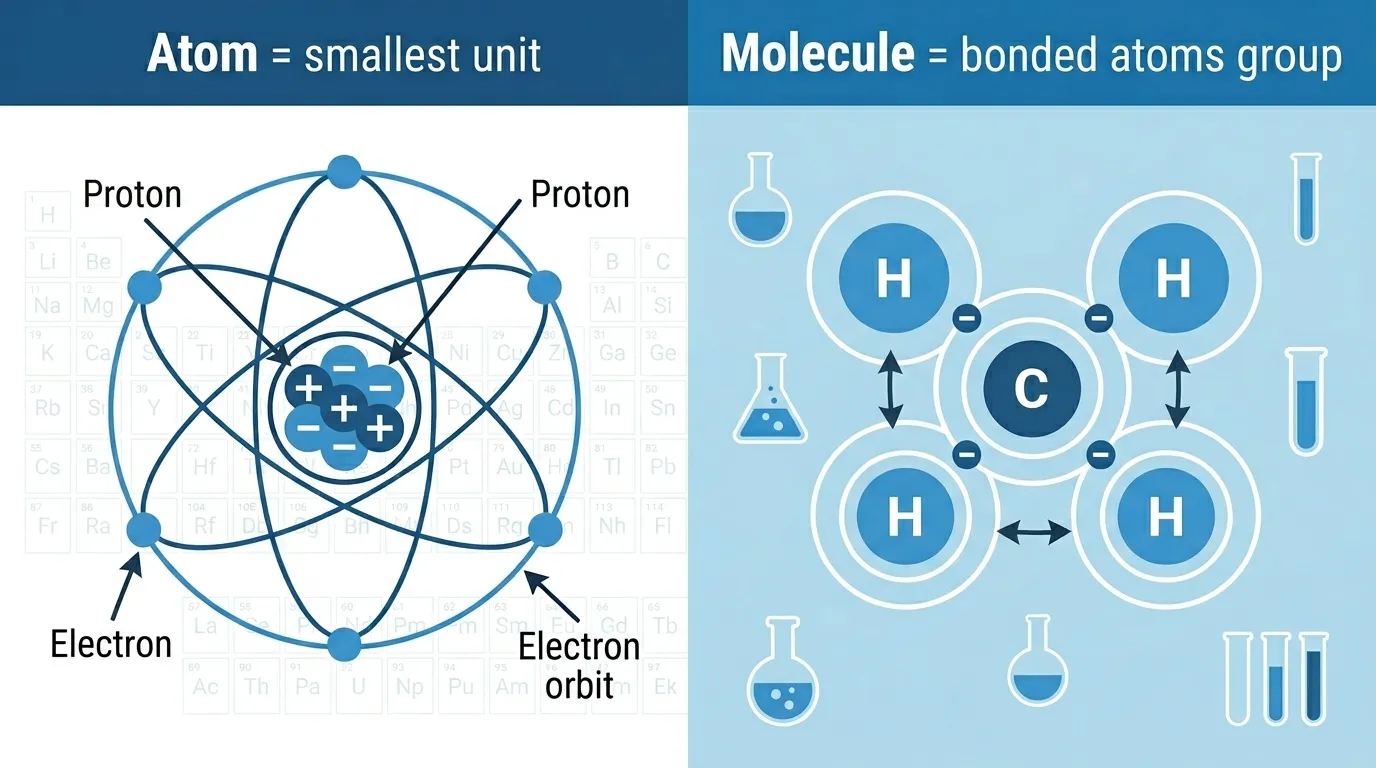
The difference between atoms and molecules is that an atom is the smallest unit of an element, while a molecule is made when two or more atoms chemically bond together.
Many students and beginners get confused about the difference between atoms and molecules because both are basic building blocks of matter. In simple words, an atom is the smallest unit of an element, while a molecule is formed when two or more atoms chemically bond together.
The difference between atoms and molecules is one of the most important concepts in chemistry, physics, biology, and modern science.
Whether you are studying school science, preparing for exams, or simply trying to understand how matter works, learning this topic clearly helps you understand everything from water and oxygen to medicines and living cells.
Today, even educational platforms and search systems like Google and YouTube recommend science content based on how users search terms like:
- “what is an atom”
- “what is a molecule”
- “atoms vs molecules”
- “difference between atoms and molecules”
In this complete guide, you will learn definitions, examples, key differences, similarities, real-world applications, common mistakes, and expert explanations in very simple language.
Difference Between Atoms and Molecules
The difference between atoms and molecules is simple:
- An atom is the smallest unit of a chemical element.
- A molecule is formed when two or more atoms join together chemically.
Real Example
A single oxygen atom is an atom, but two oxygen atoms bonded together (O₂) form a molecule.
Definition of Difference Between Atoms and Molecules
- Atom: The smallest particle of an element that keeps the chemical properties of that element.
- Molecule: A group of two or more atoms chemically bonded together.
Simple Example
Hydrogen (H) is an atom. Water (H₂O) is a molecule made from hydrogen and oxygen atoms.
Pronunciation
| Term | US Pronunciation | UK Pronunciation |
| Atom | AT-um | AT-um |
| Molecule | MOL-uh-kyool | MOL-i-kyool |
Now let’s understand these concepts more deeply in a very practical and beginner-friendly way.
Difference Between Atoms and Molecules
| Feature | Atom | Molecule |
| Meaning | Smallest unit of an element | Combination of atoms |
| Structure | Single particle | Two or more bonded atoms |
| Independence | May exist alone | Exists as bonded group |
| Chemical Bond | Not required | Required |
| Representation | H, O, Na | H₂O, CO₂, O₂ |
| Complexity | Simpler | More complex |
| Role in Matter | Basic building block | Forms substances |
| Example | One oxygen atom | One water molecule |
| Size | Smaller | Larger than atoms |
| Formation | Naturally exists | Formed by chemical bonding |
Key Differences Explained Between Atoms and Molecules
1. Atoms Are Single Units While Molecules Are Groups
The biggest difference is structure.
An atom is one tiny particle, while molecules contain multiple atoms joined together.
Real-Life Example
A single carbon atom is an atom. Glucose (C₆H₁₂O₆) is a molecule made from many atoms.
2. Molecules Need Chemical Bonds
Atoms become molecules only after chemical bonding happens.
These bonds hold atoms together in stable forms.
Example
Two hydrogen atoms bond with one oxygen atom to form water.
H2OH_2OH2O
3. Atoms Build Everything Around Us
Everything in the universe is made from atoms.
This includes:
- Air
- Water
- Food
- Humans
- Planets
Molecules are formed using these atoms.
4. Molecules Can Be Simple or Complex
Some molecules are very simple:
- Oxygen (O₂)
Others are extremely complex:
- DNA molecules
- Protein molecules
5. Atoms Represent Elements
Each atom belongs to a specific chemical element from the periodic table.
Examples:
- Hydrogen atom
- Oxygen atom
- Gold atom
6. Molecules Represent Compounds or Element Groups
Molecules may contain:
- Same atoms (O₂)
- Different atoms (CO₂)
This creates different substances.
7. Molecules Usually Determine Substance Behavior
The arrangement of atoms inside molecules affects:
- Smell
- Taste
- Color
- Reactivity
- Physical properties
Real Scenario
Diamond and graphite both contain carbon atoms, but their molecular structures differ greatly.
What Is an Atom?
An atom is the fundamental building block of matter.
Atoms contain:
- Protons
- Neutrons
- Electrons
Why Atoms Exist
Atoms exist because matter needs stable units to form physical substances.
Scientists once believed atoms could not be divided further, but modern physics discovered subatomic particles inside atoms.
Structure of an Atom
Atoms have two main regions:
1. Nucleus
Contains:
- Protons
- Neutrons
2. Electron Cloud
Electrons move around the nucleus.
Simple Analogy
Think of the nucleus as the sun and electrons as planets orbiting around it.
What Is a Molecule?
A molecule forms when atoms chemically combine.
These atoms share or exchange electrons to create stable structures.
Why Molecules Exist
Most atoms are more stable when bonded together.
That is why nature forms molecules automatically.
Example
Oxygen in the air usually exists as O₂ molecules rather than single oxygen atoms.
O2O_2O2
Types of Molecules
1. Element Molecules
Made from the same atoms.
Examples:
- O₂
- N₂
2. Compound Molecules
Made from different atoms.
Examples:
- H₂O
- CO₂
- CH₄
Difference and Similarity Between Atoms and Molecules
| Feature | Atom | Molecule | Similarity |
| Basic Nature | Single particle | Group of particles | Both are forms of matter |
| Composition | One element unit | Multiple atoms | Both involve chemical elements |
| Chemical Bonding | Not necessary | Necessary | Both participate in reactions |
| Size | Smaller | Larger | Both microscopic |
| Scientific Study | Atomic chemistry | Molecular chemistry | Both studied in science |
| Real-World Presence | Found everywhere | Found everywhere | Both build physical substances |
| Stability | Can be unstable alone | Often more stable | Both follow physical laws |
| Examples | Hydrogen atom | Water molecule | Both exist naturally |
This table clearly shows the difference and similarity between atoms and molecules for quick understanding.
How Atoms and Molecules Work Together
Atoms and molecules are deeply connected.
Simple Process
- Atoms interact.
- Electrons rearrange.
- Chemical bonds form.
- Molecules are created.
This process powers:
- Life
- Digestion
- Breathing
- Combustion
- Technology
Why Understanding Atoms and Molecules Matters
Many people think chemistry only matters in classrooms, but atoms and molecules affect everyday life constantly.
Real-World Applications
Medicine
Drug molecules interact with body cells.
Food Science
Molecular reactions create flavors and textures.
Technology
Battery performance depends on atomic interactions.
Environmental Science
Molecules like CO₂ influence climate systems.
CO2CO_2CO2
Biology
DNA molecules store genetic information.
Common Mistakes With Atoms and Molecules
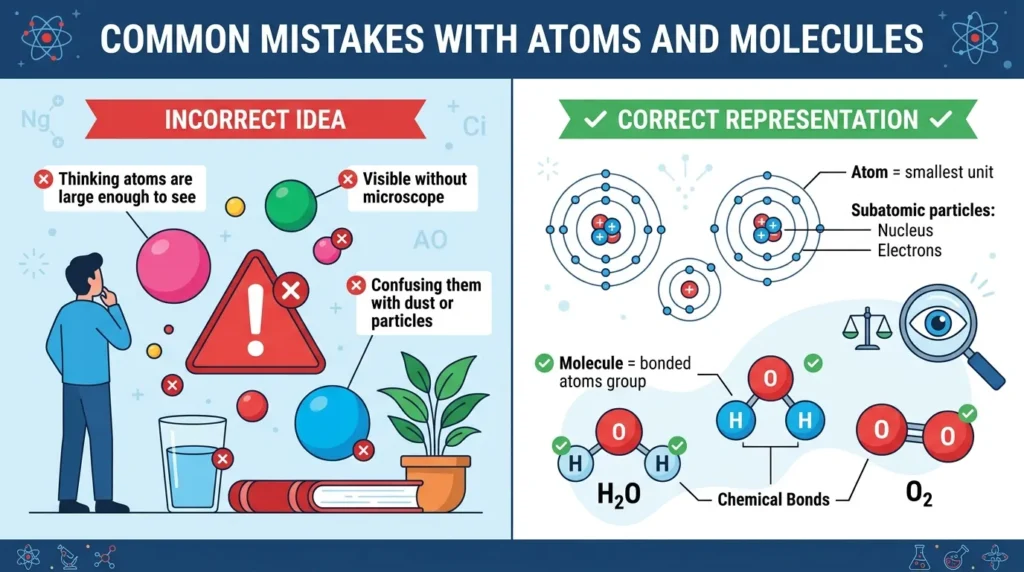
1. Thinking They Mean the Same Thing
This is the most common mistake.
Atoms are single units. Molecules are groups of atoms.
2. Assuming All Molecules Are Large
Some molecules are extremely small, like O₂.
3. Believing Atoms Are Visible
Atoms are too tiny to see with normal microscopes.
4. Confusing Molecules With Compounds
Not all molecules are compounds.
O₂ is a molecule but not a compound because it contains only one element.
5. Thinking Molecules Only Exist in Liquids
Molecules exist in:
- Solids
- Liquids
- Gases
Real Life Examples With Atoms and Molecules
Water
Water molecules contain hydrogen and oxygen atoms.
Oxygen in Air
Air contains oxygen molecules used for breathing.
Table Salt
Salt contains sodium and chlorine atoms bonded together.
Human Body
Proteins and DNA are complex molecules made from many atoms.
Smartphones and Batteries
Atomic interactions help store and transfer electrical energy.
When To Use Each Term
| Situation | Correct Term |
| Referring to smallest element unit | Atom |
| Referring to bonded particles | Molecule |
| Studying periodic table elements | Atom |
| Studying chemical compounds | Molecule |
Easy Rule
One particle = atom
Bonded particles = molecule
Why People Get Confused Between Atoms and Molecules
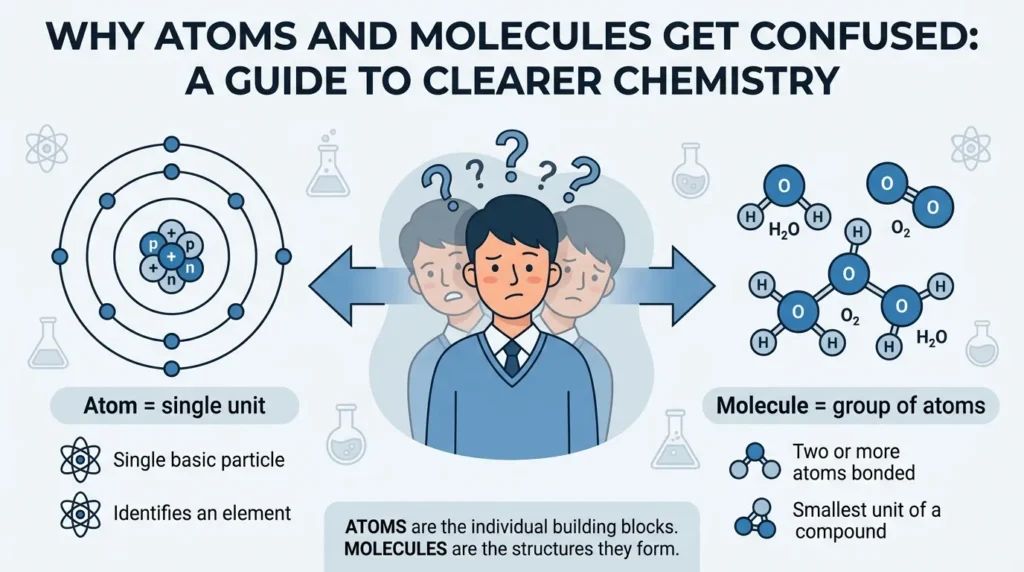
Many users confuse these terms because both are invisible microscopic particles discussed together in science education.
Main Reasons for Confusion
1. Similar Scientific Context
Both terms appear in chemistry lessons together.
2. Visual Diagrams
Science books often show atoms and molecules using similar circular models.
3. Everyday Language
People casually say “particles” for both concepts.
4. Educational Shortcuts
Some beginner explanations oversimplify the topic too much.
How Search Engines Understand Difference Between Atoms and Molecules
Search engines like Google use semantic analysis and NLP systems to understand that users searching:
- “atom vs molecule”
- “what are molecules”
- “basic chemistry concepts”
- “difference between atoms and molecules”
usually want:
- Beginner-friendly explanations
- Science education content
- Real-life examples
- Easy comparison tables
Why This Matters
Educational content ranks better when it:
- Explains concepts clearly
- Uses examples
- Answers beginner confusion
- Combines visuals and simple language
Platforms like YouTube also prioritize educational engagement signals such as watch time and topic clarity.
Expert Insight
In practical science education, students understand chemistry much faster once they realize atoms and molecules are connected rather than competing ideas. Atoms act as the raw building materials, while molecules are the organized structures formed from those materials.
Most beginners memorize definitions without understanding the deeper logic. The real insight is this:
atoms explain elemental identity, while molecules explain how substances behave in the real world.
Modern fields like nanotechnology, medicine, artificial intelligence chemistry modeling, and environmental science all depend heavily on atomic and molecular understanding.
FAQs
What is the main difference between atoms and molecules?
Atoms are single units of elements, while molecules are groups of bonded atoms.
Can one atom become a molecule?
No. A molecule needs at least two bonded atoms.
Is water an atom or molecule?
Water is a molecule made from hydrogen and oxygen atoms.
Are all molecules compounds?
No. Some molecules contain only one type of atom, like O₂.
Which is smaller: atom or molecule?
Atoms are smaller than molecules.
Can atoms exist alone?
Yes. Some atoms can exist independently.
Why do atoms bond together?
Atoms bond to become more stable chemically.
Are humans made of molecules?
Yes. The human body contains trillions of molecules built from atoms.
Conclusion
Understanding the difference between atoms and molecules is one of the most important foundations of chemistry and science. Atoms are the smallest units of elements, while molecules form when atoms chemically bond together. In simple terms, atoms are the building blocks, and molecules are the structures created from those blocks.
This concept explains how matter works everywhere around us from breathing oxygen and drinking water to using medicines, batteries, and modern technology. Atoms define the identity of elements, while molecules determine how substances behave physically and chemically.
Many beginners confuse these terms because they are closely connected, but once you understand the relationship, chemistry becomes much easier and more logical.
A simple way to remember it is:
- Atom = single particle
- Molecule = bonded group of atoms
Whether you are a student, teacher, parent, or curious learner, mastering this topic creates a strong foundation for understanding science at every level.
Read more about!
Difference Between Immigrant and Migrant: Simple Guide

I am Emily Johnson, a USA-based content writer who creates easy-to-read blogs on language and daily life topics. I explain complex ideas in simple English for students and beginners so they can understand easily.