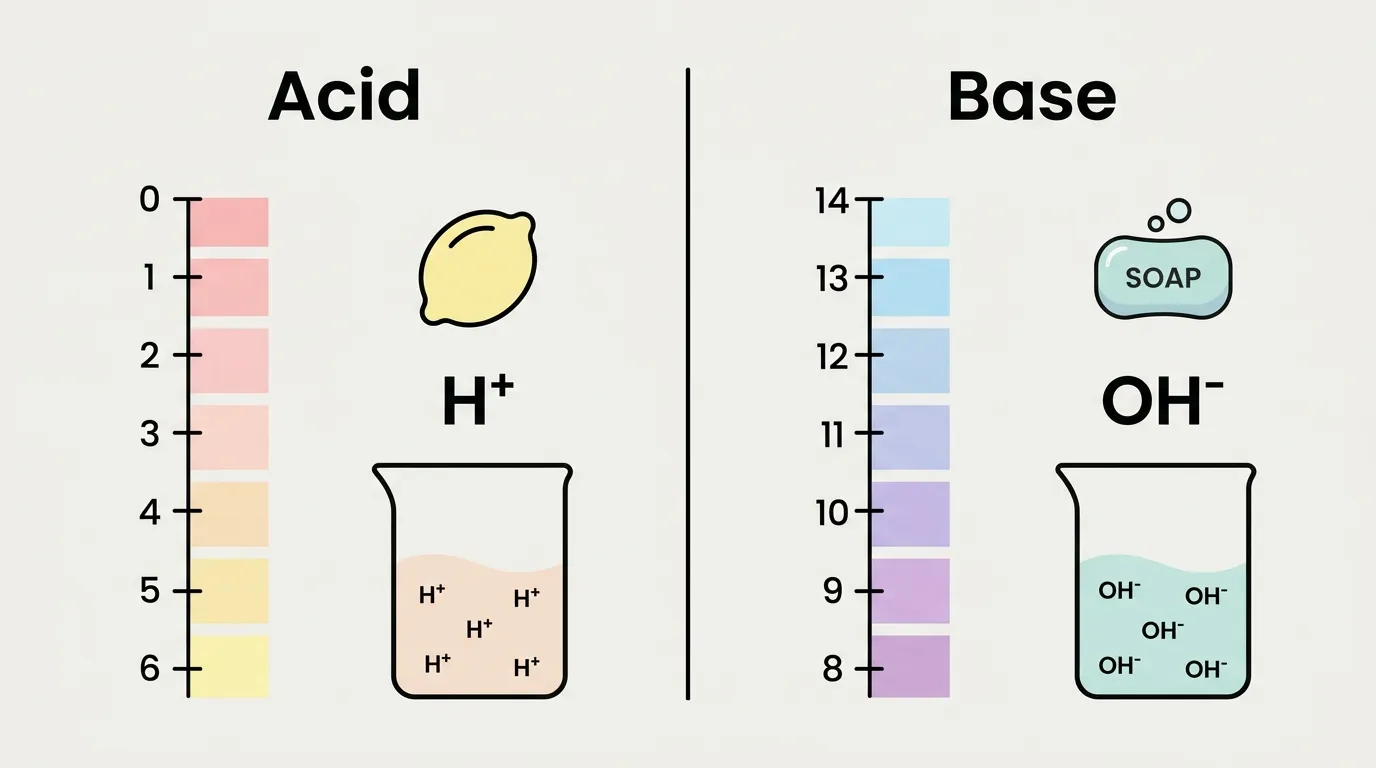
Acids donate hydrogen ions (H⁺) and taste sour, while bases accept H⁺ and feel bitter or slippery 🧪✨
Many users get confused when they search for the difference between acid and base because both are common in science, daily life, and even in food.
In simple words, acids and bases are opposite types of chemical substances but their behavior, taste, and reactions are very different.
If you’ve ever wondered what is acid and base, how they work, or why they react differently, you’re not alone. The difference between acid and base is one of the most searched chemistry topics because it appears in school, exams, and real-life situations like cleaning, cooking, and health.
In this guide, you’ll learn everything clearly:
- What acids and bases are
- How they behave
- Real-world examples
- When to use each
Let’s understand this clearly step by step.
🟢 Difference Between Acid and Base
The difference between acid and base is simple:
- Acids release hydrogen ions (H⁺) in water.
- Bases release hydroxide ions (OH⁻) or accept hydrogen ions.
👉 Example: Lemon juice is acidic, while soap is basic.
🟢 Definition of Difference Between Acid and Base
- Acid: A substance that releases hydrogen ions (H⁺) in a solution and usually tastes sour.
- Base: A substance that releases hydroxide ions (OH⁻) or accepts H⁺ ions and often feels slippery.
🟢 Pronunciation
- Acid: /ˈæs.ɪd/ (US & UK)
- Base: /beɪs/ (US & UK)
Now that you know how to say them correctly, let’s move deeper into understanding how they actually differ.
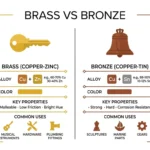
🟢 Comparison: Acid vs Base
| Feature | Acid | Base | Similarity |
| Definition | Releases H⁺ ions | Releases OH⁻ ions or accepts H⁺ | Both are chemical substances |
| Taste | Sour (like lemon) | Bitter (like baking soda) | Both interact with taste receptors |
| pH Level | Less than 7 | Greater than 7 | Both measured on pH scale |
| Feel | Can sting or burn | Slippery or soapy | Both can be harmful if strong |
| Reaction | Reacts with metals | Reacts with acids | Both react chemically |
| Examples | Vinegar, lemon juice | Soap, ammonia | Both found in daily life |
| Strength | Strong or weak acids | Strong or weak bases | Both vary in strength |
| Use | Food, digestion, batteries | Cleaning, medicine | Both used in industries |
🟢 Key Differences Explained Between Acid and Base
1. Ion Production
Acids produce H⁺ ions, while bases produce OH⁻ ions.
👉 Example: Hydrochloric acid releases H⁺, while sodium hydroxide releases OH⁻.
2. Taste Difference
Acids taste sour; bases taste bitter.
👉 Lemon is sour (acid), baking soda is bitter (base).
3. pH Scale Behavior
Acids have pH below 7, bases above 7.
👉 Water sits neutral at 7.
4. Reaction with Indicators
Acids turn blue litmus red; bases turn red litmus blue.
👉 This is a common lab test.
5. Chemical Reactions
Acids react with metals to release hydrogen gas, bases do not.
👉 Example: Acid + metal = bubbles.
6. Feel and Texture
Bases feel slippery; acids do not.
👉 Soap feels smooth because it’s basic.
7. Neutralization
Acids and bases cancel each other.
👉 Acid + Base → Salt + Water
🟢 What Is the Difference Between Acid and Base in Chemistry?
In chemistry, acids and bases exist to balance reactions. This system helps control chemical stability.
- Acids donate protons (H⁺)
- Bases accept protons
This concept comes from the Brønsted-Lowry theory, widely used in modern chemistry.
👉 In real scenarios, this helps control reactions in labs, industries, and even the human body.
🟢 Why Do Acids and Bases Exist?
Acids and bases exist because chemical reactions need balance.
- Acids help break substances (digestion)
- Bases help neutralize and stabilize
👉 Example: Your stomach uses acid to digest food, while your body uses bases to control excess acidity.
🟢 How Do Acids and Bases Work in Real Life?
Let’s look at practical usage:
- Food Industry: Citric acid adds sour taste
- Cleaning: Bases like ammonia remove grease
- Medicine: Antacids neutralize stomach acid
👉 In practical use, both are essential for daily life.
🟢 Difference Between Acid and Base in Daily Life
Many people don’t realize how often they use acids and bases:
- Toothpaste → Basic (protects teeth)
- Soft drinks → Acidic
- Soap → Basic
- Vinegar → Acidic
👉 Everyday life is full of acid-base reactions.
🟢 How Does the pH Scale Explain the Difference Between Acid and Base?
The pH scale runs from 0 to 14:
- 0–6 → Acidic
- 7 → Neutral
- 8–14 → Basic
👉 The lower the pH, the stronger the acid.
👉 The higher the pH, the stronger the base.
🟢 Difference and Similarity Between Acid and Base
| Feature | Acid | Base | Similarity |
| Nature | Proton donor | Proton acceptor | Both follow chemical laws |
| Taste | Sour | Bitter | Both interact with senses |
| Reaction | Reacts with metals | Reacts with acids | Both undergo reactions |
| pH Range | <7 | >7 | Both measured on same scale |
| Examples | Lemon, vinegar | Soap, ammonia | Both exist in daily life |
| Function | Break substances | Neutralize acids | Both maintain balance |
This table clearly shows the difference and similarity between difference between acid and base for quick understanding.
🟢 Common Errors People Make with Acids and Bases
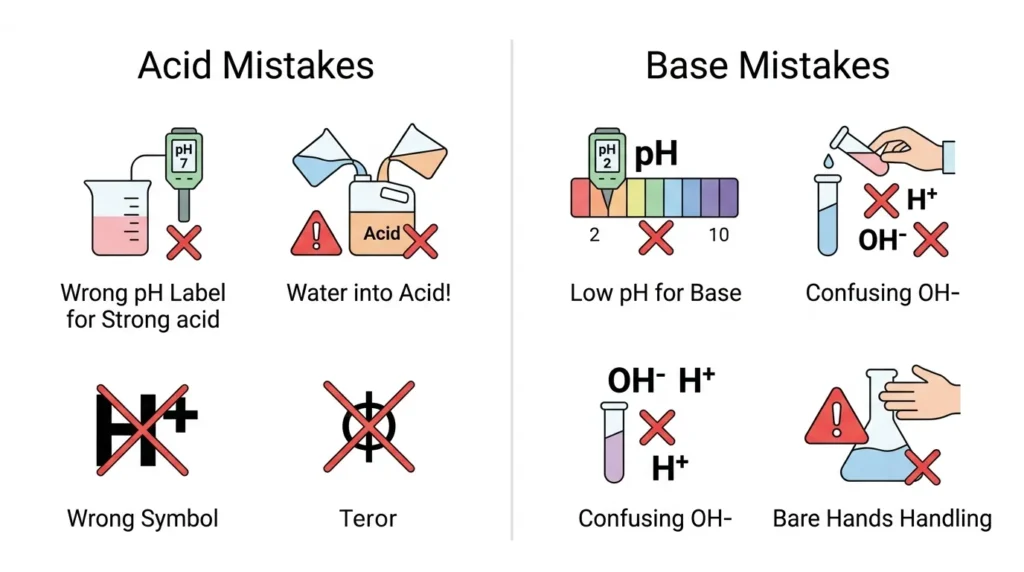
- Thinking all acids are dangerous
👉 Weak acids like lemon juice are safe. - Assuming bases are harmless
👉 Strong bases can burn skin. - Confusing pH values
👉 Lower pH = stronger acid, not base. - Mixing without knowledge
👉 Some reactions can be harmful. - Ignoring real-life usage
👉 Both are everywhere, not just labs.
🟢 Everyday Real-Life Examples of Acids and Bases
- Personal Use: Lemon juice (acid) in cooking
- Health: Antacids (base) for acidity
- Cleaning: Soap removes oil (base reaction)
- Industry: Battery acid powers cars
- Agriculture: Soil pH control
🟢When to Use Acids vs Bases
- Use acids when you need:
- Cleaning rust
- Food flavoring
- Digestion support
- Use bases when you need:
- Cleaning grease
- Neutralizing acid
- Soap production
🟢 Reasons Behind the Confusion Between Acids and Bases
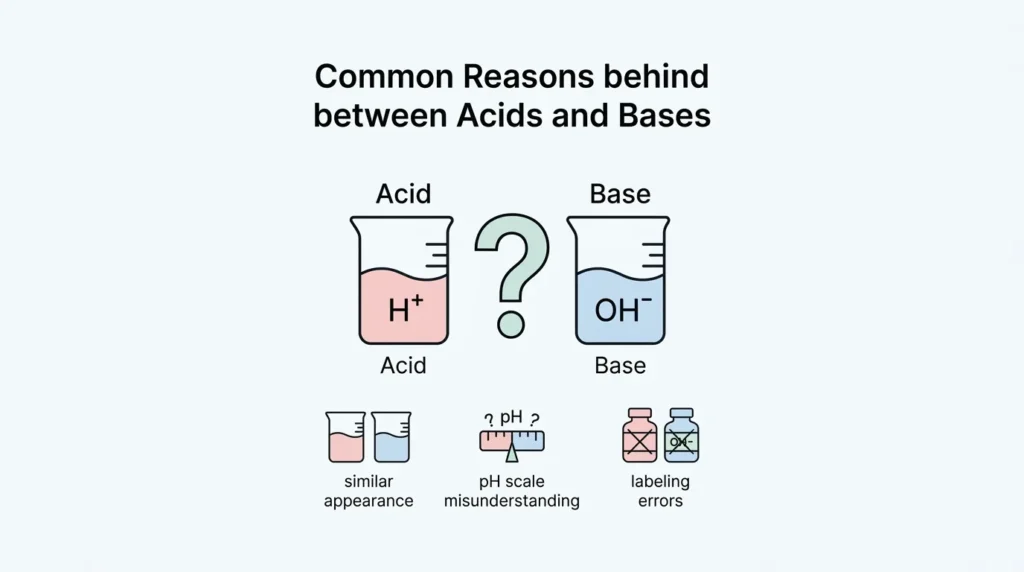
- Similar chemical terms
- Both used in daily life
- Lack of clear examples
- Misleading school explanations
👉 Many beginners think they are similar but they actually do opposite jobs.
🟢 How Search Engines Understand “Difference Between Acid and Base”
Search engines like Google analyze this query as informational + comparison intent.
They prioritize:
- Clear definitions
- Comparison tables
- Real-life examples
Platforms like YouTube and Instagram also show visual explanations because users engage more with simple comparisons.
👉 This is why structured content ranks higher.
🟢 Expert Insight
In real scenarios, understanding acids and bases is not just academic it’s practical.
From my experience explaining basic chemistry concepts, most beginners struggle because they memorize definitions but don’t understand behavior.
👉 The key insight:
Think of acids as “donors” and bases as “acceptors.”
This simple logic solves most confusion.
🟢 FAQs
1. What is the main difference between acid and base?
Acids release H⁺ ions, while bases release OH⁻ ions.
2. How can I identify acids and bases?
Use pH scale or litmus paper test.
3. Are all acids dangerous?
No, weak acids like vinegar are safe.
4. Why do bases feel slippery?
Because they react with skin oils.
5. What happens when acid meets base?
They neutralize to form salt and water.
6. Is water an acid or base?
Water is neutral (pH 7).
7. Where are acids used in daily life?
Food, digestion, batteries.
8. Where are bases used?
Cleaning, soap, medicine.
🟢 Conclusion
The difference between acid and base becomes very simple once you understand their core behavior. Acids release hydrogen ions and usually taste sour, while bases release hydroxide ions and feel slippery. These two are chemical opposites, yet they work together to maintain balance in reactions, industries, and even the human body.
In real-world use, acids help in digestion, food flavoring, and industrial processes, while bases are essential for cleaning, neutralizing, and maintaining stability. Many users get confused because both are part of everyday life, but their roles are completely different.
To remember easily:
👉 Acid = H⁺ donor (sour)
👉 Base = OH⁻ provider (bitter/slippery)
Once this concept is clear, you won’t need another article to understand the difference between acid and base.
Read more about!
Difference Between Lager and Pilsner: Clear Guide

I am James Carter, a professional content writer from the United States. I specialize in writing simple and clear comparison blogs that help students and readers understand difficult topics easily. My writing focuses on everyday language, education, science, and lifestyle topics. I believe that learning should be easy for everyone, so I use simple words and real-life examples in my articles.